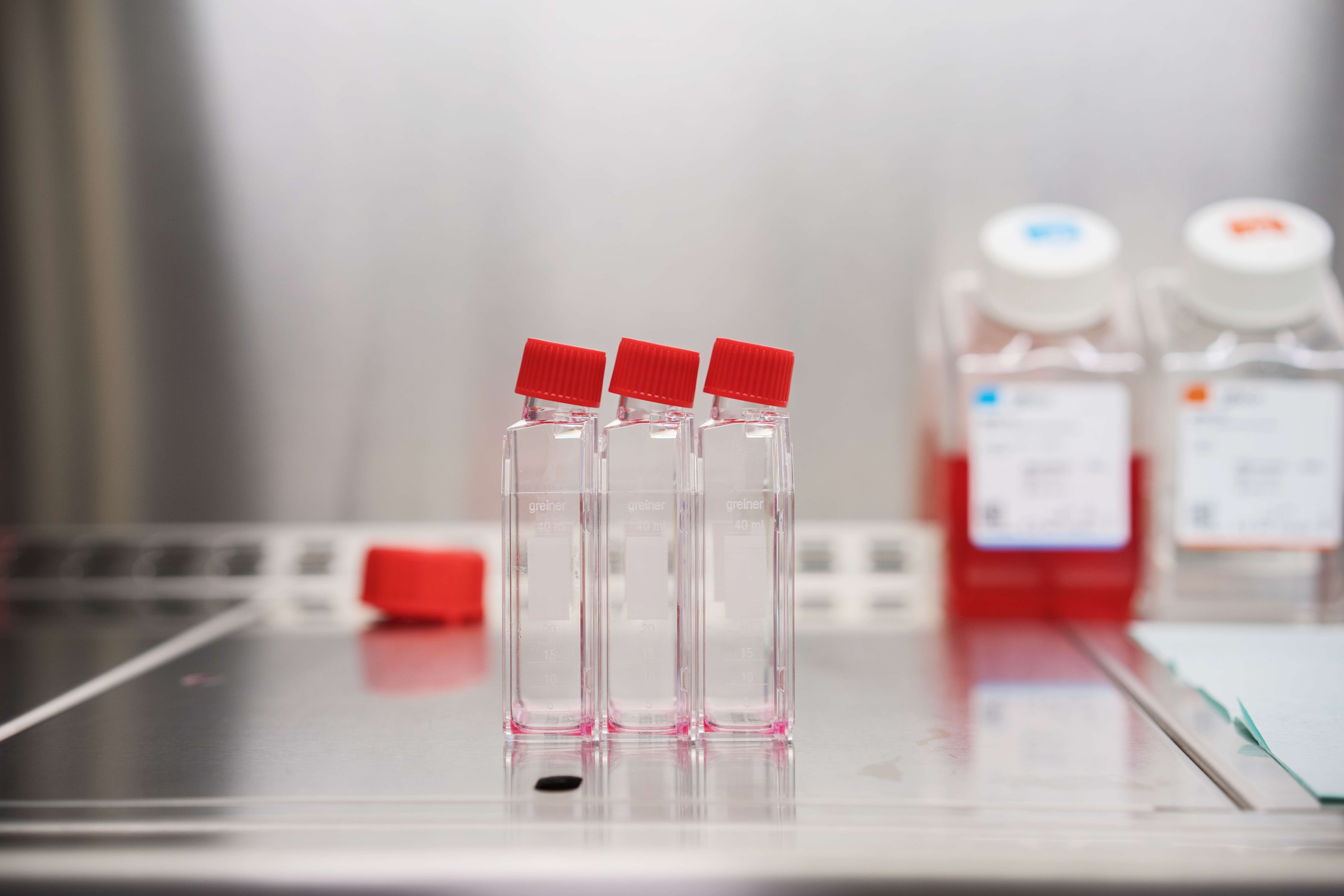
We made the next important step on the way to the registration of our drug candidates in the European Medicines Agency.
The next phase of research has started for our two medicinal products: BCX-CM-J – a drug candidate for the treatment of degenerative joint lesions and BCX-CM-AD – a drug candidate for the treatment of atopic dermatitis. It’s the safety phase called TAS (Target Animal Safety), in which the safety of the route of drag administration is tested. We sent a product batch for testing in the certified CRO (Clinical Research Organisation). The series has been manufactured in accordance with the GMP (Good Manufacturing Practice) pharmaceutical standard.
This is an important moment for us – if we confirm the safety of the route of administration of our drug candidates, we will be able to proceed to the next stage – to the tests of their effectiveness in a clinical field trial executed on clinical patients.
Puls Biznesu about our plans: Bioceltix announces that it will soon debut on NewConnect – the company has recently completed the pre-IPO round. So far, the company has obtained a total of approx. PLN 20 million in financing for the development of a still innovative method of treating pets ...
ArticleForbes about Polish start-ups in the coronavirus crisis: “The year 2020 was a big challenge for companies, individual sectors of the economy and probably all of us. In the coming weeks and months of 2021 it will be similar. The condition of start-ups is no exception here. it is easier and ...
ArticleAccording to the „Investors’ Zone”, an interesting year is being prepared on the stock exchange: “This year, an exceptionally large group of companies is choosing NewConnect. As many as 63 companies declared their willingness to enter the alternative market, and several more in the ...
ArticleGazeta Wrocławska wrote about us: “Bioceltix is working on innovative biological medicines used in the treatment of common autoimmune and inflammatory diseases in companion animals. The company from Wrocław has something to fight for. This is indicated by the numbers on the global drug ...
ArticleWe create at Bioceltix veterinary biological medicines, and in 2020 we announced plans for a stock exchange debut and we obtained a permit from the Main Pharmaceutical Inspector to manufacture veterinary biological drugs. Despite the pandemic, we continue our projects without downtime. You can ...
ArticlePuls Biznesu about our plans: Bioceltix announces that it will soon debut on NewConnect – the company has recently completed the pre-IPO ...
Read fullForbes about Polish start-ups in the coronavirus crisis: “The year 2020 was a big challenge for companies, individual sectors of the ...
Read fullAccording to the „Investors’ Zone”, an interesting year is being prepared on the stock exchange: “This year, an exceptionally large ...
Read full
